Categories
Change Password!
Reset Password!


The study investigated the effectiveness and safety of a 24-week regimen of Estetrol/Drospirenone (E4/DRSP) for endometriosis management in Japanese patients.
Estetrol/Drospirenone (15 mg/ 3 mg) combination offers effective relief from the pain associated with endometriosis and improves gynecological signs and symptoms with a promising safety profile.
The study investigated the effectiveness and safety of a 24-week regimen of Estetrol/Drospirenone (E4/DRSP) for endometriosis management in Japanese patients.
In this randomized, multicenter, double-blind, placebo-controlled trial, 162 women with endometriosis were divided into two groups: one receiving E4/DRSP orally daily for 24 days followed by 4 days of placebo, and the other receiving a daily placebo for 28 days. During the confirmatory period, the therapies were administered for six cycles (24 weeks). Alteration in visual analogue scale (VAS) scores for intense pelvic discomfort (lower abdominal and back discomfort) from baseline to after six cycles was the key outcome ascertained.
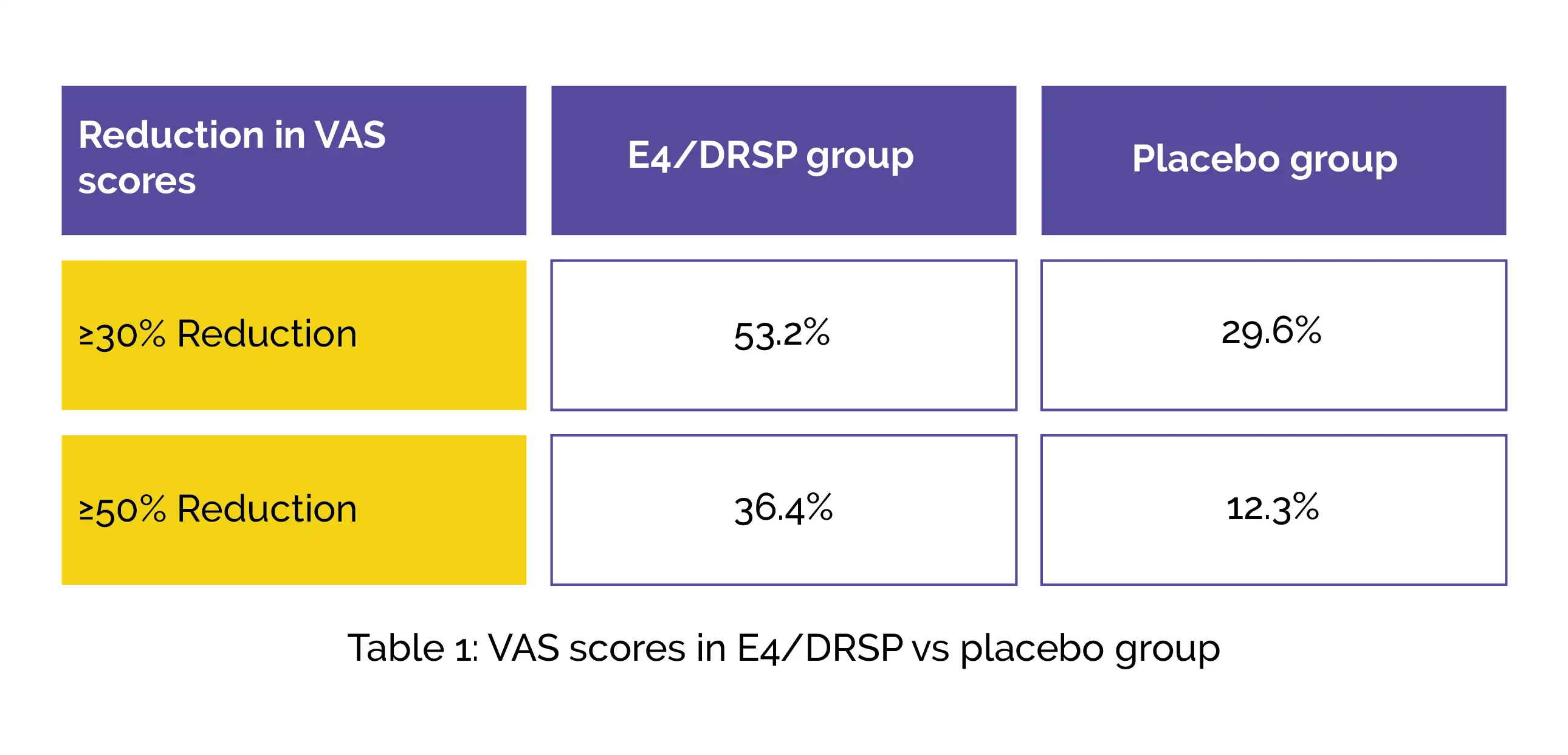
The E4/DRSP group experienced a notable reduction in pelvic pain, with an average decrease of 33.2 mm on the VAS, compared to a smaller decrease in the placebo group. Reduction in the VAS (≥30% and ≥50%) was also observed, as shown in Table 1 below:
E4/DRSP use considerably improved objective gynecological findings (induration, tenderness, and uterine mobility), reduced endometrioma size, and boosted the quality of life compared to placebo. No safety issues were noted, and there were minimal differences in hemostasis parameters between the study groups.
E4/DRSP demonstrated effective pain relief and improvement in endometriosis symptoms. It also exhibited a favorable safety profile, making it a promising novel option for treatment, with a potentially reduced risk of thromboembolic events.
Fertility and Sterility
Efficacy and safety of the combination of estetrol 15 mg/drospirenone 3 mg in a cyclic regimen for the treatment of endometriosis-associated pain and objective gynecological findings: a multicenter, placebo-controlled, double-blind, randomized study
Tasuku Harada et al.
Comments (0)